Dr. Dana
My journey from pharmacy to landmark medical breakthroughs.
Note: Cannabis Confidential shifted content strategy in 2026 with insights from trusted partners, industry insiders, and thought leaders who’ve got something to say. There will be no more paywalls or content fees; think of it as The Players Tribune for the cannabis industry, with an ethos of honesty, trust and respect.
It is with great enthusiasm and gratitude that I share my story with this audience and moreover, I want to thank Todd Harrison for facilitating this forum for the cannabinoid wellness space during what’s shaping up to be a monumental time in history.
I’m Dr. Dana Lambert (known by most as Dr. Dana) and I’m founder & CEO of a cannabinoid biopharma company called Andira Pharmaceuticals.
It’s been over the past 10 years that I’ve considered it my life’s work to develop cannabinoid-based medicine to help people heal from breast cancer, invasive surgery, chronic wounds, and antibiotic-resistant infections.
Our therapies are backed by both clinical and non-clinical efficacy data. Based on our results to date, ranging from triple synergy against superbug bacteria to clinical relief of complex surgical pain, it is tremendously gratifying to see our products emerging as landmark medical breakthroughs with potential to become first-in-class therapies for their medical indications.
This article is about my belief in cannabinoid therapeutics, why I am endeavoring to transform modern medicine with them, and the backstory on how I arrived where I am today.
A Whale of a Tale
I grew up on Vancouver Island, British Columbia, Canada, and being highly driven to succeed as a business professional, embarked on a business degree at the University of British Columbia (UBC) fresh out of high school at age 17.
Let’s just say I’ve always felt the entrepreneurial spirit. However, during my first year at UBC, I came to a self-realization that my true passion was the science of medicine.
Specifically, I was drawn like a moth to a flame to the underlying molecular chemistry and pharmacology of medicine, and how plants so frequently offer substances that act medicinally in humans.
I was allowed to transfer into the Faculty of Science at UBC, completed the pre-requisite classes, and was accepted into the Faculty of Pharmaceutical Sciences where I graduated as a licensed pharmacist (BSc Pharm) in 2010.
In pharmacy school I took elective courses on indigenous medicine and read scientific publications on plant pharmacology. I also had the privilege of learning about plant medicine with indigenous elders on Vancouver Island and in Central America.
One of the most impactful stories I recall was how a woman with esophageal cancer was cured by drinking tea prepared by an indigenous elder from Pacific yew tree bark.
This was after failing medical treatment (chemotherapy) with the anti-cancer drug that is derived from yew tree bark (paclitaxel). The tea prepared with whole bark worked, whereas conventional medical treatment with only one active ingredient from the yew tree was ineffective.
Key takeaway: Pharmaceuticals can fail to capture the full medicinal aspects of plants, as they typically contain one active ingredient only; whereas plants often contain countless pharmacologically active ingredients.
^ not all of them necessarily contribute to a particular medicinal effect of the plant, and some of the active ingredients may have undesirable side effects.
Before I returned to the UBC Pharmaceutical Sciences Faculty to dive headfirst into developing cannabinoid therapeutics while completing a PhD, I worked for the Vancouver Island Health Authority and Vancouver Coastal Health Authority doing clinical research and quality & safety projects.
I then worked as a hospital pharmacist on Vancouver Island. My pharmacy career lasted 2.5 years and it was a wakeup call.
I saw firsthand at patients’ bedsides the suffering, and too often, the mortality, caused by cancer, treatment-resistant infections, non-healing wounds, and chronic pain.
My job was to optimize drug therapy for patients, but all too often, the available treatments fell short. I feel immense gratitude to have interacted with each of my patients, and for them helping me to better understand human disease states, and both the science and the art that is medicine.
I am also grateful to have learned about healthcare systems, what factors affect the prescribing of medications, including what contributes to a medication becoming “standard-of-care” and highly lucrative for the company producing it.
During pharmacy school, we were taught by a medicinal chemistry professor about the endocannabinoid system and cannabinoid drugs (both phytocannabinoids and cannabinoid analogues) circa 2009.
I often referred to those course notes, while also seeing mounting publications on cannabinoid pharmacology—and it didn’t take long for me to realize the gargantuan potential of cannabinoid medicine.
Given their proven pharmacology and potential to help address a countless diversity of medical conditions that I had treated firsthand, for which available drug therapies often failed or were non-optimal, I knew that if someone could develop cannabinoid therapeutics properly, it could be a huge win for modern medicine and humanity.
The entrepreneur in me also saw a tremendous business opportunity, albeit it one that would require utmost dedication from those involved to be successful.
Understanding modern healthcare systems and medical treatment paradigms, I knew that the only way for a cannabinoid therapy to have a strong impact in the real world would be to have it developed and approved along the same regulatory pathway as all other medicinal products which have an approved medical use indication.
This would ensure that it could be available and prescribed in mainstream medicine, with both patients and healthcare practitioners having assurances of the product’s efficacy and safety for its labeled use. This is what I meant earlier by properly.
I left my career as a pharmacist in 2013 and entered cannabinoid research and development at UBC, by engaging in the Graduate Studies program in the Faculty of Pharmaceutical Sciences where I would eventually receive my PhD in cannabinoid pharmacology and chronic pain pathophysiology.

I developed novel small-molecule cannabinoid analogue drugs that could selectively activate the CB2 cannabinoid receptor and then compared their activity to known phytocannabinoids and other natural compounds.
I also did a primary chronic pain pathophysiology study to determine whether the inflammatory cells in the brain and spinal cord could be targeted therapeutically with CB2-activating drugs for fibromyalgia and other chronic musculoskeletal pain conditions.
^ my study indicated that yes, this appeared to be the case, at least in a pre-clinical research model.
During this time I started Andira Pharmaceuticals with the help and support of Entrepreneurship UBC (e@UBC), the University-Industry Liaison Office, several rockstar mentors from the biopharmaceutical industry whom I met via UBC, and my longtime family friend and former employer Michael Wall, who co-founded and helped launch the company with a seed investment from a group of his family and friends.
Andira Pharmaceuticals started with a small laboratory operating in the UBC Faculty of Pharmaceutical Sciences with one full time scientist and we conducted early-stage discovery research under the purview of various Professors across the UBC campus.
Then we reached our first major discoveries, filed patents, and acquired exclusive worldwide rights to the technologies.
This evolved into a situation whereby Andira contracted further R&D to optimize our discoveries and generate product candidates that could advance to the clinical trials.
These studies were conducted by a range of academic institutions (including UBC), research institutes and hospitals, and other leading contract research organizations (CROs), located both inside and outside of Canada.
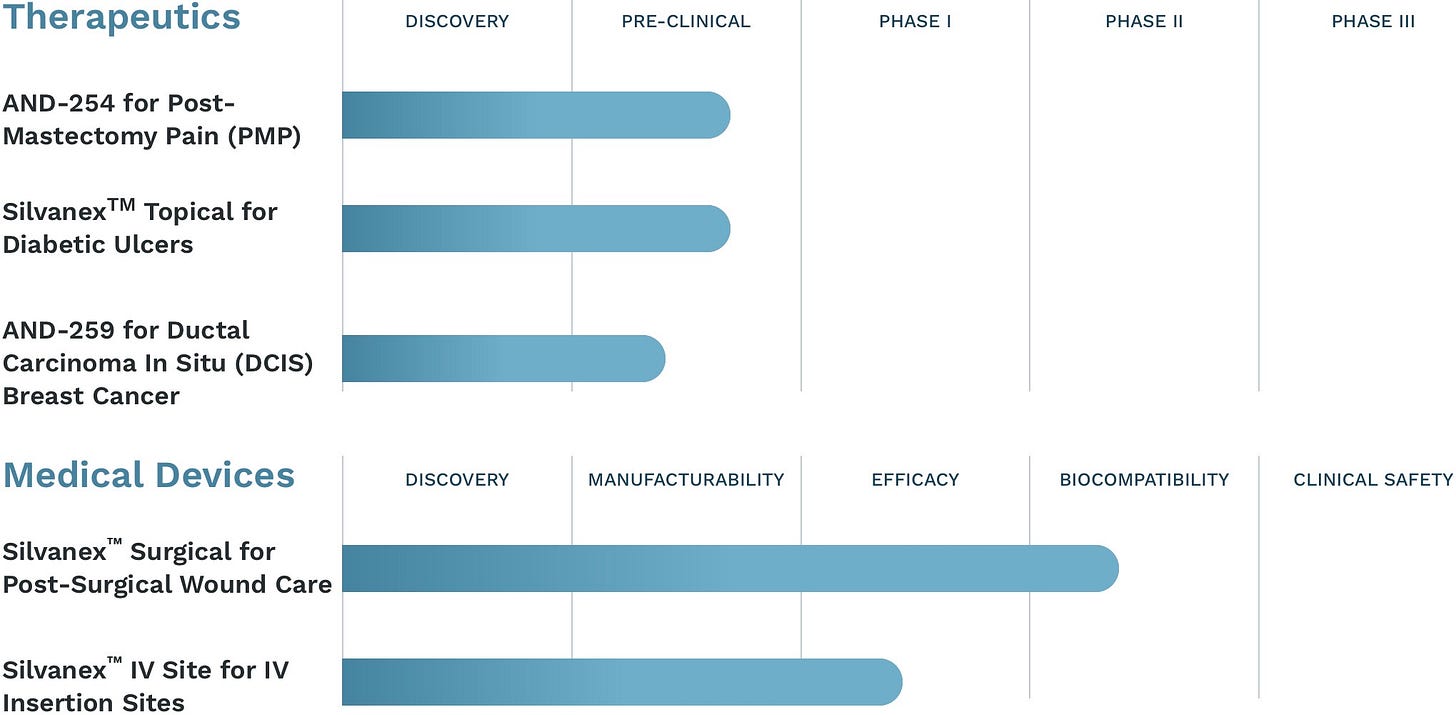
Today, I’m proud to say that we have a therapeutic product pipeline of three drug candidates, two of which are bridging forward into clinical trials.
In addition to our product candidates, we have a patent-protected drug delivery platform technology specific to cannabinoids which I developed together with a colleague, and a patent-protected infection control technology, which has market use applications far beyond what our product pipeline can accommodate.
For this reason, we are actively licensing our platform technologies to third party companies, whereby Andira will benefit tremendously from their success, and in turn will allow us to stay focused on achieving success with our therapeutic products.
Three Phases of Leaves
I believe in the cannabinoid therapeutics Andira is developing for three main reasons.
For one, cannabinoids, within certain dosing parameters and exposure levels, are generally highly safe and non-harmful to people. Hence, I believe that cannabinoid therapeutics which are designed and made with care, including stringent quality control measures, have little potential to cause harm.
Secondly, the cannabinoid therapeutics we are developing have strong efficacy data, which we have generated through both conventional pre-clinical drug testing, and via pilot clinical case studies. The triple synergy against superbug bacteria and clinical relief of complex surgical pain that I noted above are but a glimpse of the full picture.
Efficacy is where most (cannabinoid) therapeutics have historically failed during development: oftentimes products have been unable to meet clinical endpoints in Phase II or III trials, because it is not accomplishing what it was intended to do medicinally in real people.
I cannot wait for our upcoming clinical trials, where I believe our patients will reap significant benefit and our shareholders will have cause for celebration.
The third reason why I believe in our cannabinoid therapeutics, at the risk of sounding highly unconventional, is that I followed the recommendation of my indigenous elder teachers before embarking on product development: I asked the cannabis plant (yes, I asked the plant itself, as recommended by the elders) to create better medicine.

I believe that the cannabis plant should be held in the highest regard, with the same sanctity that we place on human health and wellness given the wealth of medicine and healing potential that it has offered us.
Hence my sincere belief in our cannabinoid therapeutics is weighted heavily on the understanding that both human and plant intelligence came together to create them.
There are more people than I could possibly thank and acknowledge here who have contributed to the success of Andira: My academic and industry mentors, our research team, our executive team, our company advisors, our world-leading service providers, company co-founder and fellow Director Michael Wall, who, together with Dr. Ethan Russo, fellow Director and Chief Medical Officer, have provided the highest level of service to the company, and finally, every one of our incredible shareholders who have backed the company and allowed our success in advancing better medicine to become a reality.
You are all a continuous source of inspiration that allows me to do what I do every day with purpose and gratitude.
Andira’s Silvanex™ Shows Zero MRSA Resistance Potential in Landmark AMR Study
This is third-Party content and does not reflect (or not not reflect) the views of Canna Confidential or CB1 Capital. CB1 Capital has a position in Andira Pharma.
Dr. Dana Lambert is an entrepreneur and biopharmaceutical executive with a background in healthcare management and pharmaceutical research. Dr. Lambert founded Andira Pharmaceuticals and led the discovery and development of each product candidate in the Andira Pharmaceuticals pipeline, with over 25 patents pending globally, a US patent granted, multiple peer-reviewed publications and several research awards in pharmacology and natural product innovation. She holds a PhD in Pharmaceutical Sciences from the University of British Columbia and started her career as a hospital pharmacist and healthcare executive based in British Columbia, Canada.